CGKB News and events Management strategies
Fungi - faba bean
Contributors to this section: ICARDA, Syria (Siham Asaad, Abdulrahman Moukahal).
Ascochyta Blight, Leaf blight, Pod spot
Scientific names
Ascochyta fabae Gossen, Sheard, C.J. Beauch. & Morrall [anamorph], (1986): Didymella fabaeG.J. Jellis & Punith, (1991) [teleomorph].
Significance
Yield losses of 10-30% can occur in seasons favorable for the disease. Discoloration of seed can seriously reduce its market value.
Symptoms
Symptoms occur on leaves, stems and pods of infected plants and can be confused with the early stages of chocolate spot. On leaves, small, circular, dark-brown spots appear first. As the disease develops, they enlarge and turn light and then change to dark grey in color. They become irregular in shape, often zonate, and may coalesce to cover most of the leaf surface. Leaf tissue next to the lesions may become black and necrotic. Within the lesions, numerous pinhead- sized black fruiting bodies (pycnidia) of the fungus develop. These appear only under moist conditions and are often concentrically arranged on the stem; lesions are more elongated, sunken and darker than leaf lesions and are usually covered with scattered pycnidia. Stems may split and break at the point of infection causing plants to lodge. On pods, lesions are sunken and have pale centre and dark margins; they can be covered by numerous pycnidia. Well developed lesions can penetrate the pod and infect developing seeds causing them to be shrunken and discolored. Badly infected seeds have yellowish brown stains on the outer seed coat, which considerably reduces its market value.
Ascochyta blight can cause seed staining in pods close to maturity even when disease levels in the crop have been too low to warrant fungicide sprays. Faba bean seed that has greater than 25% seed coat discoloration can reduce the emergence of seed by 30%. Seed that has less than 5% seed coat discoloration will usually have normal levels of germination.
Hosts
Faba bean.
Geographic distribution
Africa (Egypt, Morocco); America (Argentina, Canada (NS); Asia (China, Syria, Israel, Japan, Korea, Turkey); Australasia (Australia, New Zealand); Europe (Britain, Czechoslovakia, Denmark, Germany, Italy, Jersey, Norway,Russia).
Biology and transmission
This fungal disease has an asexual and sexual stage; the asexual stage is most common. In this stage the fungus survives mainly on infected seed and on crop residues.
Spores of the fungus produced on crop residues can be carried onto new crops by wind. Infection can occur at any stage of plant growth, provided conditions are favorable. Moisture is essential for infection to occur.
During wet weather, the disease can spread further than in dry conditions because spores of the fungus are carried onto neighboring plants by wind and rain splash.
Detection/indexing method in place at the CGIAR Center at ICARDA
- Malt Extract Agar
Treatment/control
Crop rotationc
- Since only Faba bean is susceptible to ascochyta blight caused by A. fabae inclusion of non-host crops in the rotation will bring about a reduction in the inoculum level.
- Faba beans should not be grown more frequently than every 3-4 years.
- Furthermore, new crops should not be grown near fields that were infested with blight in the previous year due to the risk of infection by air borne ascospores.
Tillage
- Under most conditions, deep ploughing will hasten decomposition of infected Faba bean straw and remove it as a source of inoculum. Studies conducted in the Pallouse Region in Washington State showed that the pathogen survived in naturally infected plant material for more than 2 years when situated on the soil surface, but lost its viability rapidly at soil depths of 10-40 cm. All infested crop residues and volunteer plants should therefore be destroyed by thorough tillage and proper crop rotation.
Fungicide application
- Growers should take special care to protect green and healthy foliage during pod filling, especially if blight symptoms appear and increase in severity. Tests with application of Bravo 500 at different crop stages have shown that spraying at early podding reduced pod damage and markedly increased seed yields in the moderately resistant cultivars. Foliar fungicide applications may not prove cost effective when disease pressure is low. Thiram-based fungicides are registered for the treatment of grey mould infected seed. While seed treatment can improve establishment, it does not provide any protection from air-borne infection later in the crop.
Procedure followed at the centers in case of positive test
- Destroying seed for intensive spots, and seed treatment for no visible spots
References and further reading
Ascochyta Leaf and Pod Spot of Faba Bean [online]
CABI. Ascochyta fabae. [Distribution map] [online].
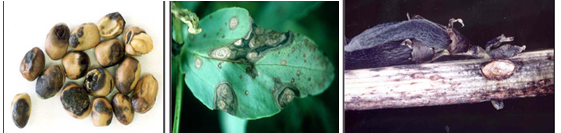 Ascochyta blight (photos:dpi.vic.gov.au) |
Scientific name
Botrytis fabae Sardina
Other scientific name
Botryotinia fabaeWu & Lu (1991).
Significance
Loss depends on the severity of infection, the time at which infection occurred, and the amount of spring rainfall. In unprotected crops, the disease can reduce yields by at least 30-50% under conditions favorable for disease development. In addition, seed from badly affected plants may have a reddish-brown discoloration, which lowers its market value.
Symptoms
These are varied, and range from small spots on the leaves to complete blackening of the entire plant. Leaves are the main part of the plant affected, but under favorable conditions for the disease it also spreads to stems, flowers and pods. Two stages of the disease are usually recognized. First, a non-aggressive phase, when discrete reddish-brown spots are "peppered" over the leaves and stems, and then an aggressive phase, when spots darken in color and coalesce to form larger grey-brown target spots that may eventually cover the entire plant. Small black sclerotia may sometimes be found inside the stems of badly diseased plants.
Red-legged earth mite (RLEM) damage can be mistaken for chocolate spot. This starts as silvery patches which become red-brown, similar in color to chocolate spot but form large irregularly-shaped areas. Red-legged earth mite damage usually occurs during the seedling stage and on the lower leaves.
Factors such as phosphorus or potassium deficiency, water logging and excessive weed burdens which reduce crop vigor may make plants more susceptible to the development of chocolate spot. Leaves which have been damaged by insect attack or in wheel tracks are also more susceptible.
Hosts
Vicia spp. especially Vicia faba (field or broad bean); also reported from Phaseolus beans, pea and lentil.
Geographic distribution
Europe, Middle East, N.E. and S. Africa, S.E. Asia, Australasia, S. America. More recent records are from Korea, India, S. Australia, Canada and Norway.
Biology and transmission
Chocolate spot, caused by Botrytis fabae and Botrytis cinerea can survive either as sclerotia in the soil or on crop debris, in infected seed, or on self-sown volunteer plants. In hitherto unaffected areas the disease often becomes established by the sowing of infected seed. In subsequent years, the initial infection usually occurs when spores formed on old bean trash are carried by wind into new crops. These spores may move long distances. Once the disease becomes established it rapidly spreads within a crop, and within 4-5 days of infection spores can be formed on infected tissue and initiate secondary spread of the disease. The fungus is most aggressive under cool, humid conditions, particularly at flowering time. Aggressive development of stem infection late in the season can cause the crop to lodge.
Air-borne conidia are readily produced on dead bean foliage during wet weather. The fungus perennates in bean debris and as sclerotia in the soil and is also seed-borne.
Detection/indexing method in place at the CGIAR Center at ICARDA
- Agra Plate Test
Treatment/control
- Use of clean seed is advocated. Seed treatment with fungicides such as benomyl (0.2-0.1%) chlorothalonil (0.2-0.3%) or thiabendazol (0.1-0.2%) is recommended to control the seed- borne infection. Practice a 2-3 year crop rotation along with deep plowing to reduce soil-borne inoculum.
- Foliar spray using chlorothalonil (2-3 L\ha) as single application at early bloom to early pod set provides the best protection and increases seed yield.
Procedure followed at the centers in case of positive test
- Destroying serious infected seed, and seed treatment for light infection.
References and further reading
CABI. Botrytis fabae. [Descriptions of Fungi and Bacteria]. [online]
Richardson H, Horsham DPI. 2009. Chocolate Spot of Faba Bean. [online]
 Chocolate spot (photos:dpi.vic.gov.au) |
Botrytis grey mold of faba bean
Scientific name
Botrytis cinerea Pers. 1794
Other scientific name
Botryotinia fuckeliana (de Bary) Whetzel
Significance
The disease is capable of causing serious yield losses in years when spring rainfall is high and/or there are prolonged wet periods especially if it is founded with B. fabae.
Symptoms
All aboveground plant parts of Faba bean can be affected by botrytis grey mould. Depending on the location of the crop, symptoms may initially appear either on flowers and pods, or lower in the crop canopy. The most damaging symptoms become apparent after the crop has reached canopy closure and a humid microclimate is produced under the crop canopy. The disease appears first as discrete cream colored lesions on lower leaves. These enlarge and coalesce to infect whole leaflets which later senesce and fall to the ground. Unlike ascochyta blight, no small, black fruiting bodies (called pycnidia) can be seen within the lesions. If conditions remain conducive for disease, that is warm and wet under the crop canopy for at least 4 days, infection can spread to the lower stems. These lesions will girdle the stem and become covered with a furry layer of grey mould, eventually causing stem death and whole plant death, often occur before the onset of flowering and pod fill. Infection will continue to spread resulting in patches of dead plants within crops. Pods which become infected will be covered in a grey moldy growth, rot, and turn brown when dried out.
Hosts
Botrytis cinerea has a broad crop host range collectively, including faba bean, chickpea, field pea, lupin and pasture legumes such as lucerne and clover. Other host species include a wide range of ornamental and horticultural crops.
Geographic distribution
Cosmopolitan.
Biology and transmission
The fungal pathogens Botrytis cinerea that cause botrytis grey mould can survive as several forms, these include in infected seed, sclerotia in the soil, in old infected trash, and on alternate host plants. Sowing seed that is infected by the botrytis grey mould pathogens can give rise to infected seedlings and the appearance of seedling blight symptoms, which can reduce seedling survival and reduce crop establishment. Old infected trash is an important source of fungal inoculum. Spores are produced on old trash and are carried by the wind into new crops where infection can occur.
Detection/indexing method in place at the CGIAR Center at ICARDA
- Agar Plate Test
Treatment/control
- Use of clean seed is advocated. Seed treatment with fungicides such as benomyl (0.2-0.1%) chlorothalonil (0.2-0.3%) or thiabendazol (0.1-0.2%) is recommended to control the seed- borne infection. Practice a 2-3 year crop rotation along with deep plowing to reduce soil-borne inoculum.
- Foliar spray using chlorothalonil (2-3 L\ha) as single application at early bloom to early pod set provides the best protection and increases seed yield.
Procedure in case of positive test
- Seed treatment
References and further reading
Scientific name
Colletotrichum lindemuthianum (Sacc. & Magnus) Briosi & Cavara.
Other scientific name
Colletotrichum truncatum (Schwein.) Andrus & Moore.
Significance
Not Significant
Symptoms
Leaf lesions and premature leaf drop: In most faba bean crops, the first symptoms of anthracnose appear before flowering, when the plants have 8 to 12 nodes on the main stem. This is also the time when the first tendrils form, and approximately a week before flowers start to open. If there is a large amount of inoculum in the field the first symptoms may appear earlier. Tan coloured lesions of variable size develop on the lower leaflets and the most severely affected leaflets die and drop to the ground. This premature leaf drop indicates that anthracnose may become a problem, and fungicide application should therefore be considered.
Stem lesions: Lesions on stems develop soon after the appearance of leaf lesions, primarily at the base of the plant. Stem lesions may be small, brownish with a black border, or larger, stretching along the stem. As the season progresses, more and more lesions develop at the stem base, as well as on the upper part of the stems, and many stems are girdled.
Wilt: Anthracnose causes defoliation and stem girdling, which inhibits utilization of water and nutrients, and causes the faba bean plants to wilt. As a result, large areas of brown and dying plants can be found in the field.
Hosts
The fungus Colletotrichum truncatum causes anthracnose in lentil and only attacks plant species belonging to the Lens and Vicia family, such as fababean and wild vetches. This means that a few weedy species in Canada can harbor the disease, but other crops, except fababean, are not at risk.
Geographic distribution
It is reported from Bangladish, Canada, Ethiopia, Morocco, and Syria. It is economically important only in Canada.
Biology and transmission
Small, pinhead sized fungal structures (microsclerotia) form on the infected plant tissue. They may be seen with the unaided eye in the centre of stem lesions or more easily with a hand lens (10-15 x magnification). Each microsclerotia consists of a few hundred cells with thick, black cell walls that protect the fungus from colonization by other microorganisms. Microsclerotia enable the fungus to survive between faba bean crops either on the plant debris or free in the soil. They remain viable longer when buried in the soil by tillage than left exposed to weather extremes on the soil surface.
Detection/indexing method at ICARDA
- Agar Plate Test
Treatment/control
- Use either certified seed, approved seed, or seed known to have a long disease-free history. The use of disease-free seed is the most important control measure.
- Do not plant beans for at least two years in land that has carried an infected crop.
- Remove diseased plants, where practical, to help check the spread of disease.
- Avoid cultivating and harvesting an affected crop when wet to prevent the spread of spores.
- Do not pack lightly diseased pods as anthracnose can develop during transport
- Bravo 500 (50% cholorothalonil, Zeneca Agro). The recommended rate is 0.8 - 1.6 L/acre (2.0-4.0 L/hectare) with a maximum of two applications in a season. The water rate is 90-640 L/acre (220-1600 L/hectare). Bravo must be present on the plant surface prior to the onset of fungal infection. It sticks well to the plant surface and resists removal by rain. A second application 10-14 days later may be necessary under wet weather conditions and to protect new growth. Fungal resistance to cholorothalonil has not been detected.
Procedure followed in case of positive test
- Seed treatment
References and further reading
Minchinton E, Knoxfield. 1999. Anthracnose of beans. [online]
North Dakota State UNiversity. Anthracnose Disease In Lentils. [online]
 Anthracnose (photo:paridss.usask.ca) |
Phoma blight, Spring black stem, Leaf spot
Scientific name
Phoma medicaginis var. medicaginis Malbr. & Roum, 1886.
Other scientific name
Phoma medicaginis
Significance
Not Significant
Symptoms
Small, dark brown to black dots on leaves, petioles and stems;
Leaf spots enlarge, coalesce forming irregular blotches;
Infected leaves turn yellow, wither and drop;
Black tissue may appear near base of stems;
Crown and root rot may occur; and
Infected seed pods and seed may discolour and shrivel.
Hosts
All legumes and specially alfalfa.
Geographic distribution
Cosmopolitan
Biology and transmission
Favoured by cool and wet conditions; overwinters on dead stems and leaves or in crowns and roots; spores released during periods of wet cool weather; spores spread by rain splash, wind blown or carried by insects; and new shoots exposed through infected residue.
Detection/indexing method at ICARDA
- PDA
Treatment/control
- Use certified seed; use disease resistant cultivars; crop rotation; and crop rotation; and spring burning when severe.
- Application of benomyl seed treatments (0.1 and 0.5% w/w) resulted in only a 4-5 week delay in the onset of Phoma black stem symptoms.
Procedure followed at the centers in case of positive test
- Seed treatment
References
Manitoba Agriculture, Food and Rural Initiatives. Management of Diseases of Alfalfa Seed. [online]
Fusarium wilt, Fusarium root rot
Scientific names
Fusarium oxysporum, F. moniliforme, F. equiseti, F. tricinctum and F. solani. [anamorphs]
Other scientific names
Gibberella avenacea, Gibberella moniliformis, Gibberella intricans, Gibberella tricincta, Gibberella zeae.[Teleomorphs]
Significance
The diseases cause up to 100% yield loss under heavy infestation, depending on relative humidity, soil moisture, and soil temperature.
Symptoms
The disease appears in the field in patches at both seedling and adult stages. Seedling wilt characterized by sudden drooping followed by drying of leaves and whole seedling, and apparently healthy looking roots with reduced proliferation.
The adult wilt symptoms first appear at flowering to late pod-filling stage and are characterized by sudden drooping of top leaflets of the affected plant, leaflets closure without premature shedding, dull green foliage color followed by wilting of whole plant or individual branches, apparently healthy looking roots system with a slight reduction in the lateral roots which are as difficult to pull out as the healthy plant, and no internal discoloration of the vascular system in most cases.

Fusarium |
Hosts
Widespread.
Geographic distribution
Worldwide.
Biology and transmission
If a field becomes infested with either of the wilt fungi, it remains so indefinitely. The causal fungi can be carried from one field to another on farm equipment, on lentil refuse, and in wind- or waterborne soil. They may be introduced into new areas on the seed. Once introduced into a field it may take two years or more for the fungi to increase in numbers where an appreciable amount of disease is evident. A soil temperature of (23° to 27°C) is most favorable for Fusarium or true wilt, and a slightly higher optimum temperature (27°C) for near wilt. Race 5 of the near wilt fungus, not found in Illinois, infects at lower temperatures. The wilt-producing fungi are soil inhabitants that penetrate the lentil plant through the root hairs and fibrous roots. They grow upward through the stem, often well into the upper branches, in the water conducting tissue (xylem). This process interferes with the passage of water from the roots to the stems, leaves, and pods resulting in yellowing, dwarfing, and wilting of plants. The Fusarium fungi do not reproduce on living plants but produce large numbers of microscopic spores (microconidia, macroconidia, and chlamydospores) in and on dead stems and roots. The spores are splashed or blown about within fields. The spores germinate and the resulting hyphae penetrate the host. The fungi survive in soil for 10 years or longer, in the absence of a lentil crop, as thick-walled chlamydospores. Survival is related to the association of the Fusarium fungi with the roots of nonhost crops. The fungi are also capable of infecting seeds.
Detection/indexing method at ICARDA
- Blotter Test
Treatment/control
- Choose resistant varieties when available. Remove stricken growth and sterilize clippers (one part bleach to 4 parts water) between cuts.
- Control garden insects, such as cucumber beetles, which are known to spread the disease.
- Remove all weeds from the garden (many weed species host the disease). The biological fungicide Mycostop will control wilt caused by Fusarium. If the disease persists, it is best to remove the entire plant and solarize the soil before planting again.
- To solarize the soil, you must leave a clear plastic tarp on the soil surface for 4-6 weeks during the hottest part of the year. Soil solarization will reduce or eliminate many soil inhabiting pests, including nematodes, fungi, insects, weeds and weed seeds.
Procedure in case of positive test
- Rotation, seed treatment
References and further reading
Economic Plants and their Diseases, Pests and Weeds. Diseases: Fusarium Root Rot and Fusarium Wilt. [online]
PAN Germany, OISAT. Fusarium wilt. [online]
Planet Natural. Fusarium wilt. [online]
Stem rot, Sclerotinia crown rot and root
Scientific name
Sclerotinia sclerotiorum (Lib.) de Bary, (1884)
Other scientific name
Hymenoscyphus sclerotiorum(Lib.) W. Phillips, (1887).
Significance
Sclerotinia sclerotiorum infects nearly 400 plant species and causes economic damage to a wide range of crops.
Symptoms
Signs and symptoms caused by S. sclerotiorum vary depending on the host. However, the most obvious sign is the appearance of white fluffy mycelial growth that will later produce sclerotia. The fungus infects and produces mycelium at the base of the plant that will eventually move up the stem causing it to rot.
Sclerotinia sclerotiorum on seedlings (collar rot) produces small black-brown lesions at the base of the stem. These will eventually cause rapid destruction of the seedlings as the fungus moves up the stem and into the leaves. Fruits such as cucumber, squash, and eggplant are also susceptible to S. sclerotiorum. In this case the fungus causes a wet rot that will eventually engulf the entire fruit. White mycelium can be seen both externally and within the infected fruit.
 Sclerotinia disease (photo: www.igzev.de) |
Hosts
Sclerotinia sclerotiorum is among the most nonspecific, omnivorous, and successful of plant pathogens. Plants susceptible to this pathogen encompass 64 families, 225 genera, and 361 species.
Geographic distribution
Sclerotinia sclerotiorum has a world-wide distribution; however it is most prevalent in cool moist regions.
Biology and transmission
Approximately 90% of the life cycle of Sclerotinia species is spent in soil as sclerotia. At certain times of the year, depending on the inherent nature of the fungus and various environmental factors, the sclerotia germinate and form either mycelium which can infect a host, or an apothecium. Infection of host plants by mycelium can occur at or beneath the soil-line. Sclerotia germinate in the presence of exogenous nutrients and produce hyphae which invade nonliving organic matter, forming mycelium which then infects living host tissues. Penetration of the host cuticle is achieved by mechanical pressure. Mycelium infection is unlikely to infect plants located more than 2 cm from a sclerotium.
Apothecium produced by Sclerotium releases ascospores that disseminate by air. Generally, 10-20 C is the optimum temperature range for carpogenic germination. Under favorable conditions, including adequate moisture, ascospores germinate within 3-6 hours of release. In one study, apothecia collected from the field discharged ascospores continuously for seven days in the laboratory. Ascospores infect nonliving host tissues, germinate, and inundate the nonliving plant part with mycelium. Then, the fungus invades healthy plant tissues with mycelium. After the plant or plant part dies, sclerotia are formed either on or in the plant. The sclerotia return to the soil for a "resting" period (which can be weeks or years) before they become active, which requires the appropriate environmental conditions.
Sclerotia are the structures which allow species of Sclerotinia to survive for long periods of time under adverse conditions. The black, melanized rind appears to act as a protectant from invasion by microorganisms. Soil temperatures, pH, and moisture appear to have little direct effect on their survival, though the combination of high temperatures and high moisture appears to encourage the degradation of sclerotia near the soil surface.
Air-borne ascospores are the most important means of spread. Mycelium from sclerotia can also cause infection, but usually this mode of infection stays within an area. Moving contaminated soil (on farm equipment, shoes, infected seedlings) and fertilizing with manure from animals fed infected plant debris, are two common ways of spreading sclerotia or mycelium from place to place. Irrigation also has been shown to be involved in the spread of Sclerotia spp. from field to field. Irrigation transported sclerotia remained viable for at least 10-21 days in flowing water. In addition, seed may be an infective source, either from contaminating sclerotia or internal mycelium. However, support for this method of transmission is questionable.
Detection/indexing method at ICARDA
- PDA , blotter test
Treatment/control
- There are essentially three methods of controlling Sclerotinia sclerotiorum; chemical, cultural, and biological control. Chemical and cultural control is most widely used, while biological control is used to a lesser degree. The only chemical control that is employed to control S. sclerotiorum is fumigation. The soil is fumigated to reduce the amount of inoculum present. Cultural practices are often used to prevent the spread of the pathogen from one host to another by killing the pathogen or by providing unfavorable conditions for the pathogen to develop. Sanitation, proper ventilation, and adequate spacing of plants are all effective cultural practices used by growers in controlling S. sclerotiorum. Although biological control is used less extensively than chemical and cultural methods, some biological control agents have been labelled. Many antagonistic fungi such as Coniothyrium minitans, Gliocladium roseum, Gliocladium virens, Sporodesmium sclerotivorum, and Trichoderma viride have been shown to provide effective control of S. sclerotiorum. These mycoparasitic fungi inhibit the growth of S. sclerotiorum by destroying existing sclerotia while inhibiting the formation of new sclerotia. All three methods for the control of S. sclerotiorum are important to long term health and viability of the plants affected by this pathogen. Addition of compost and organic fertilizers can decrease disease levels. Fungicides such as methyl thiophanate, PCNB (pentachloronitrobenzene) and chlorothalonil are also effective. Consult label directions and carefully follow instructions for application.
- Removing the debris after harvest will reduce the amount of inoculum in the soil. In some cases, alternation with a nonsusceptible crop also reduces inoculum level.
Procedure in case of positive test
- Rotation and seed treatments
References and further reading
http://www.extento.hawaii.edu/Kbase/Crop/Type/s_scler.htm
http://www.cals.ncsu.edu/course/pp728/Sclerotinia/S_sclerotiorum.html
http://www.igzev.de/englisch/projekte.php?abteilung=4
Scientific names
Stemphylium botryosum Rotem,Y. Cohen & I. Wahl, (1966) [Anamorph]:
Pleospora herbarum(Pers.) Rabenh., (1854) [Teleomorph]
Significance
It is reported to be seedborne
Symptoms
Spot-causing fungal disease occurring in leaf. The lesions are at first water-soaked small spots and then become blackish brown lesion divided by leaf veins. The lesions often extend from the leaf rim, fuse with the adjacent lesion, and become large. The leaf shrinks and withers in results. The diseased leaf defoliates at the early stage. The species of the causal organism is same with leaf spot fungi of alfalfa, but the pathogenicity is differentiated.
Hosts
On a wide range of hosts including apple, ash, broad bean, clover, endive, gladiolus, gramineae, lettuce, lupin, muskmelon, onion, Onobrychis, Medicago sativa, mangold, tomato, Trifolium, Vicia.
Geographic distribution
Worldwide; very common in temperate and sub-tropical regions.
Biology and transmission
Survival: The fungi are carried-over on infected plant debris (leaves and stems), seed and in the soil.
Environmental conditions: Stemphylium leaf spot may develop under warm moist conditions.
Dispersal: Spores of these fungi are spread by wind and rain splash. Generally air-borne as ascospores or conidia. These penetrate the leaf or petiole via stomata. May also occur on seeds and in soil.
Detection/indexing method at ICARDA
- Blotter test, agar plate method
Treatment/control
- Seed treatment. These diseases are usually not a problem after the end of the rainy season except in Valleys where they can cause damage up to harvest under conditions of high humidity and heavy night time dew. (ICARDA).
Procedure followed in case of positive test
- Seed treatment
References and further reading
CABI. Pleospora herbarum. [Descriptions of Fungi and Bacteria]. [online].
Clarke R, Knoxfield. 1999. Diseases of lucerne - 2: Fungal leaf diseases. Agriculture Notes. [online].
Scientific names
Rhizoctonia solaniJ.G. Kühn 1858 [Anamorph]
Thanatephorus cucumeris(A.B. Frank) Donk 1956 [Teleomorph].
Significance
Not Significant.
Symptoms
R. solani primarily attacks below ground plant parts such as the seeds, hypocotyls, and roots, but is also capable of infecting above ground plant parts (e.g. pods, fruits, leaves and stems). The most common symptom of Rhizoctonia disease is referred to as "damping-off" characterized by non germination of severely infected seed whereas infected seedlings can be killed either before or after they emerge from the soil. Infected seedlings not killed by the fungus often have cankers, which are reddish-brown lesions on stems and roots. In addition to attacking below ground plant parts, the fungus will occasionally infect fruit and leaf tissue located near or on the soil surface. This type of disease often occurs because the mycelium and/or sclerotia of the fungus are close to or splashed on the plant tissue.
Although most Rhizoctonia diseases are initiated by mycelium and/or sclerotia, several important diseases of beans, result from basidiospore infection. These basidiospores also serve as a source for rapid and long distance dispersal of the fungus. The basidiospores germinate to produce hyphae that infect leaves during periods of high relative humidity and periods of extended wet weather. Under these conditions, basidiospores can often be observed on the base of stems near the soil surface or on the underside of leaves in the plant canopy.
The pathogen is transported in infested soil or through movement of diseased plants or bean pods. Potential for seed-borne inoculum also exists.
Although basidiospores are wind-borne, their role in initiating disease has not been considered important except for foliar diseases (web blight) in high humidity.
Hosts
R. solani is a very common soilborne pathogen with a great diversity of host plants.
Geographic distribution
Common throughout the world.
Biology and transmission
Rhizoctonia solani produces thread-like growth called hyphae ; large masses of hyphae are referred to as mycelium. The hyphae of Rhizoctonia solani have the following characteristics, some shade of brown, a special type of cross wall within the hyphae, called a dolipore septum, each cell is multinucleate (has many nuclei) rather than binucleate, branches that are produced at right angles, no asexual spores are formed by the mycelium.
In general, the growth rate of Rhizoctonia solani is very rapid and a typical isolate can grow across a 90 mm petri plate in three days. Small, oval cells produced in branched chains or clusters are formed. These are called monilioid cells and have slightly thicker walls than the mycelium. Large aggregates of these cells are called sclerotia which are black to brown and 3 to 5 mm long.
The hyphae of Rhizoctonia solani have many nuclei (commonly 4 to 8) per cell. This distinguishes it from similar fungi that have only 2 nuclei per cell. Those fungi with hyphal characteristics similar to Rhizoctonia solani, but with only 2 nuclei per cell, are called binucleate types and are generally non-pathogenic. These Rhizoctonia solani-like fungi are saprophytic, do not cause disease, and feed on dead organic matter.
Following invasion of the host by Rhizoctonia solani, sexual spores are formed on specialized structures called basidia. Four spores are produced on each basidium. Basidia are formed when the environment is moist and sufficient growth of the fungus has occurred. Basidiospores are wind-dispersed and germinate with moisture. Each basidiospore has a single nucleus. The hyphae produced by germinating spores will fuse (anastamose), forming new hyphae with a mixture of different types of nuclei.
Observations of basidiospores on diseased host tissue are not common. Several methods have been developed to produce basidiospores in pure culture but it remains difficult.
In recent years, the recognition of different anastomosis groups has helped to elucidate the complexity of this large fungal group. Having no asexual spores and only the few morphological characteristics to define "Rhizoctonia solani isolates", many fungi were placed into this species. However, it is now recognized that Rhizoctonia solani has multinucleate cells and other related fungi (with 2 nuclei per cell), the binucleates, do not belong in that species. Further, when isolates are paired, only related mycelium will fuse and thus anastomosis groups have been defined by a set of tester isolates. A set for Rhizoctonia solani isolates and another set for binucleate Rhizoctonia solani-like fungi are also available.
Detection/indexing method at ICARDA
- Agar Plate Test
Treatment/control
- Obtain high quality seeds and avoid seeds that may be contaminated with the pathogen. In fields known to have Rhizoctonia solani, prepare planting areas to increase drainage and prevent water accumulation. Set plants with adequate spacing to avoid crowding and formation of high humidity in the foliage.
- Addition of compost and organic fertilizers can decrease disease levels. Fungicides such as methyl thiophanate, PCNB (pentachloronitrobenzene) and chlorothalonil are also effective. Consult label directions and carefully follow instructions for application.
- Removing the debris after harvest will reduce the amount of inoculum in the soil. In some cases, alternation with a nonsusceptible crop also reduces inoculum level.
Procedure followed in case of positive test
- Rotation, seed treatment
References and further reading
http://www.extento.hawaii.edu/Kbase/Crop/Type/r_solani.htm#MANAGEMENT
http://www.cals.ncsu.edu/course/pp728/Rhizoctonia/Rhizoctonia.html
 Rhizoctonia root disease (photos:www.cals.ncsu.edu) |
Viruses - faba bean
Contributors to this section: ICARDA, Syria (Siham Asaad, Abdulrahman Moukahal).
Scientific name
Bean Yellow Mosaic Virus(BYMV)
Other scientific names
Bean virus 2, Canna mosaic virus ,Gladiolus mosaic virus, Gloriosa stripe mosaic virus.
Significance
During pulse surveys from 2000-2005, BYMV infections were uncommon and found in less than 1% of surveyed crops. However, BYMV was sometimes found in faba bean crops and occasionally in peas with within crop virus incidences of 1-15% and up to 7% respectively. In NSW, a small survey showed that faba bean crops had an average within crop incidence of BYMV of 26% of plants (ranging from 1-63%) (van Leur et al. 2002). Field surveys in 1998-1999 showed that some faba bean and field pea crops were infected with BYMV and the within crop virus incidences were 1-31% and 1-11% respectively. Plot trials showed that lupins infected with the necrotic strain of BYMV can have grain yields reduced by 95%.
Symptoms
Leaves of infected plants show mild mosaic followed by narrowing. The new growth from leaf axils has leaves that are narrow, elongated and light green. Early infections adversely affect plant growth and yield.
Lentils develop mild mosaic, light green or yellow leaves, reduction in leaf size and stunting may occur. Infected plants produce very little seed.
Narrow-leaf lupins, infected with the necrotic strain of BYMV initially develop yellow leaves followed by necrosis of growth tips and plant death. Non-necrotic strains of BYMV cause yellowing and dwarfing but do not cause death of the plant.
Subterranean clover plants develop leaf mottling, leaf deformation and distinct yellowing between the veins. Plants become dwarfed and symptoms usually occur in patches and along the edges of paddocks.
Hosts
The host range of BYMV is wide and not limited to Fabaceae. The virus is reported to infect nearly 200 species in 14 families. Temperate pulse hosts include chickpeas, faba beans, field peas, lentils and lupins. Temperate legume pasture hosts include lathyrus, lucerne, vetch and medic and clover species. BYMV has a number of sub-tropical and tropical pulse hosts, including soybeans, peanuts and French beans as well as legume pasture hosts. It also infects ornamental hosts, the most common being gladiolus species.
Geographic distribution
Belarus, Belgium: Bulgaria, Czech Republic,Denmark, Finland, Former Yugoslavia, France, Germany, Greece, Hungary, Lithuania, Netherlands, Poland, Portugal, Romania, Russian Federation, Russia (Europe),Russian Far East, Spain, Sweden, Ukraine, United Kingdom, China, Georgia (Republic), India , Indonesia, Iran, Israel, Japan, Jordan, Kazakhstan, Korea, Lebanon, Pakistan, Syria, Turkey, Uzbekistan, Egypt, Kenya, Libya, Morocco, South Africa, Sudan, Tanzania, Tunisia Zambia, Zimbabwe, Western Hemisphere, Canada, Chile, Dominican Republic, Jamaica, Mexico, Montserrat, Peru, USA, Alabama, Kentucky, Tennessee, Australia, New Zealand.
Biology and transmission
BYMV is transmitted by more than 50 aphid species in a non-persistent manner: the main species are Acyrthosiphon pisum, Aphis fabae, A. gossypii, Aulacorthum solani Brevicoryne brassicae, Myzus persicae and Rhopalosiphum maidis. During surveys in Victoria the following BYMV vectors in pulse crops were found : Acyrthosiphon kondoi, Aphis craccivora, Aulacorthum solani, Brevicoryne brassicae and Myzus persicae. Acyrthosiphon kondoi, Aphis craccivora, Myzus persicae, Brachycaudus rumexicolens, Lipaphis erysimi, Rhopalosiphum maidis, R. padi, Sitodion miscanthi and Therioaphis trifolii have been reported as BYMV vectors.
The virus is also transmitted through seed of most temperate pulses, including faba beans, field peas, lentils, and lupins and through seed of a number of forage legumes and clovers. In Victoria, 18% of lentil seed lots tested had BYMV infections of 0.1-0.9%. In WA, the following BYMV seed transmission is reported: yellow and white lupins 3-6%, field peas 0.3-0.8%, faba beans 0.4%, lathyrus 0.1-0.2% and vetch 0.5%.
Seed transmission of BYMV in medics and clovers has also been reported in WA as follows: Melilotus indica (0.5%), Medicago polymorpha (0.9%), M. truncatula (0.3%), M. indica (1%), Trifolium arvense (0.1%), T. campestre (0.2%) and T. glomeratum (0.05%).
Detection/indexing method at ICARDA
- TBIA (Tissue Blot Immuno Assay test), Enzyme-linked immunosorbent assay (ELISA)
Treatment/control
- Seed is considered to be one of the main sources of BYMV, therefore sowing virus tested seed is recommended and commercial seed tests are available.
- BYMV causes heavy losses in narrow leafed lupins, so only virus tested seed is recommended for sowing. Virus resistant lupin varieties are now available.
- BYMV-infected pastures are another major source of the virus, which is then spread to crops by aphids. Chemical control of aphids is not an effective method for controlling non-persistently transmitted viruses such as BYMV.
- Pulse crops should be sown away from legume pastures to minimise the spread of BYMV.
- The spread of virus can also be reduced by controlling weed hosts from in and around paddocks.
Procedure followed at the centers in case of positive test
- Destroying seed, production healthy seed
References and further reading
Temperate Pulse Viruses: Bean Yellow Mosaic Virus (BYMV)
http://www.staff.kvl.dk/~thluj3/symptoms/fababymv.html
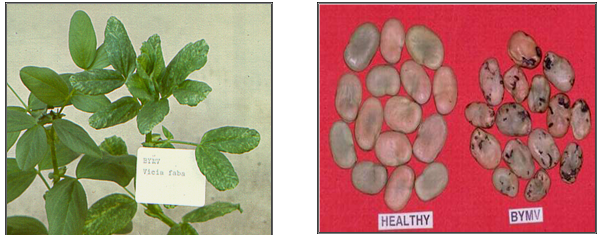 BYMV (photos:www.staff.kvl.dk and www.dpi.vic.gov.au) |
Scientific name
Broad Bean Stain Virus (BBSV).Lloyd, Smith & Jones, 1965.
Other scientific names
Broad bean F1 virus, Broad bean Evesham stain virus
Significance
Nineteen genotypes of lentil were studied in Syria for variability in seed transmission rates and yield losses induced by broad bean stain virus (BBSV) infection. Seed transmission rates of BBSV varied between 0.2% and 32.4%, whereas yield loss due to infection varied between 14% and 61%.
Symptoms
The disease is characterized by a mild mottling on the leaves.
The affected plants are reduced in growth, which is easily recognized, especially when they are compared with healthy plants. Yield reduction of 15-16% could occur in some different cultivars. Seed from infected plants occasionally show dark staining on the seed.
Hosts
Hosts restricted to Leguminosae; inoculated 44 species in 19 dicotyledonous families, but the virus infected only 7 species of legume: Crotalaria spectabilis, Lupinus hirsutus, Melilotus alba, Phaseolus vulgaris (French bean), Pisum sativum (pea), Trifolium incarnatum (crimson clover) and Vicia faba (broad bean).
Geographic distribution
Europe, Austria, Hungary, Italy, Poland, United Kingdom, Asia (as a whole), China, Syria, Turkey, Africa (as a whole) Egypt.
Biology and transmission
Transmission by Vectors: Transmitted by a vector; an insect; Apion vorax, Sitona spp.; Coleoptera.
Transmission through Seed: Common in certain varieties of broad bean, transmitted to up to 10% of the progeny of infected plants.
Transmission by Dodder: Not tested.
Virus transmitted by mechanical inoculation; transmitted by grafting; transmitted by seed (up to 10% in some Vicia faba cultivars).
Detection/indexing method at ICARDA
- Tissue Blot Immuno Assay test (TBIA), Enzyme-linked immunosorbent assay (ELISA).
Treatment/control
- For the three seed-borne viruses (BBSV, PSbMV and CMV) it is important to avoid the use of virus-infected seed lots.
- Rogue early infected plants to prevent further disease spread.
- For the aphid-transmitted viruses, spray systemic aphidicides, especially in lentil nurseries or seed multiplication plots. This should be considered in situations where winged forms of aphids begin to appear. Spraying will help suppress aphid populations in lentil fields.
- For BBSV, which is transmitted by weevils, treat seed with promet (12 ml/kg seed) to help reduce the vector and hence disease spread.
- All these viruses have a wide host range including other legumes such as dry peas, phaseolus bean, faba bean, alfalfa and clover.
Procedure in case of positive test
- Destroying infected seed, producing healthy seed
References and further reading
http://www.dpvweb.net/dpv/showdpv.php?dpvno=029
http://image.fs.uidaho.edu/vide/descr112.htm#Taxonomy
http://www.icarda.org/Publications/Field_Guides/Lentil/Lent7.Html
http://www.cababstractsplus.org/abstracts/Abstract.aspx?AcNo=19916777532
http://www.padil.gov.au/pbt/index.php?q=node/20&pbtID=152
 BBSV (photos:www.padil.gov.au) |
Scientific name
Pea Seed-borne Mosaic Virus (PSbMV)
Other scientific names
Pea fizzle top virus, Pea leaf rolling virus, Pea leaf roll mosaic virus, Pea leaf rolling mosaic virus.
Significance
PSBMV is of economic importance in pea, faba bean and lathyrus, mainly due to its effect on seed quality. It has been mistakenly considered to be a minor disease because it often causes only minor yield loss and mild symptoms, However, at the International Centre for Agriculture in the Dry Areas (ICARDA), Syria, glasshouse studies on yield losses due to PSBMV in chickpea, faba bean, lentil and pea showed losses of 66%, 40.5%, 44.6% and 49.2% respectively. Surveys of pulse crops over a number of years in Vic and SA showed that up to 15% of crops were infected with PSBMV, with a within crop incidence of up to 5% of plants. In surveys in WA in 1999, PSBMV was found in 42% of field pea crops and the level of virus detected in pea seed stocks was up to 63%. The surveys indicated that PSBMV has a severe effect on seed quality of pulse crops with seed coat symptoms evident on >80% of faba bean, >50% of lathyrus species and field peas and 5% of chickpea seed.
Symptoms
The disease is characterized by a mild mottling on the leaves, which is not easily recognizable because of the small size of the lentil leaf.
The affected plants are reduced in growth, which is easily recognized, especially when they are compared with healthy plants. Yield reductions of 15-16% could occur in some different cultivars. Seeds from infected plants occasionally show dark staining on the seed.
Lentils may show no symptoms or there may be chlorosis in new shoots, mottling on leaves, shoot tip necrosis and stunting of plants.
Hosts
The natural host range of PSBMV is limited to the Fabaceae. It infects temperate pulses (chickpea, faba bean, field pea, lentil) other legumes (garden pea, narbon bean) and pastures (lathyrus and vetch). A number of PSBMV pathotypes have been recognised by their ability to infect a number of pea differential genotypes.
Geographic distribution
Belgium, Bulgaria, Czechoslovakia (former -), Denmark, Finland, France, Germany, Netherlands, Poland, Romania, Russia (Europe), Sweden, Switzerland, United Kingdom, China, Taiwan, India, Israel, Japan, Jordan, Lebanon, Nepal, Syria, Turkey, Egypt, Ethiopia, Libya, Morocco, South Africa, Sudan, Tanzania, Tunisia, Zambia, Zimbabwe, Western Hemisphere, Brazil, Canada, Peru, USA, Washington Australia , New Zealand.
Biology and transmission
The virus is believed to have spread worldwide through the exchange of infected seed. Seed transmission rates of up to 100% in peas and up to 44% in lentils have been reported. In Victoria, we have detected PSBMV at low levels in some commercial chickpea seedlots (0.4% of seed) and at higher levels in field pea and lentil seedlots (greater than 2% of seed). In the USA, 3% PSBMV infection in pea seedlots and 32-40% in lentil seedlots have been reported. At ICARDA, PSBMV was found to be transmitted through lentil seeds at rates of up to 44%. PSBMV is also transmitted in a non-persistent manner by more than 20 aphid species and by mechanical means. The most efficient vector is the pea aphid (Acyrthosiphon pisum). The other species are as follows: A. pelargoni, A. sesbaniae, Aphis craccivora, A. fabae, A. gossypii, A. nasturtii, Aulacorthum circumflexum, A. solani, Brevicoryne brassicae, Cryptomyzus ribis, Dactynotus escalantii, Macrosiphum avenae, M. euphorbiae, M. pisi, M. rosae, Metopolophium dirhodum, Myzus persicae, Ovatus crataegarius, Phorodon cannabis, Rhopalosiphum padi and Semiaphis dauci. In Victorian pulse crop surveys, we have found cowpea aphid (Aphis craccivora), foxglove aphid (Aulacorthum solani), and green peach aphid (Myzus persicae), which are all vectors of PSBMV.
Detection/indexing method in place at the CGIAR Center at ICARDA
- (TBIA) Tissue Blot ImmunoAssay test, Enzyme-linked immunosorbent assay (ELISA)
Treatment/control
- Seed is considered to be the main source of PSBMV, therefore sowing virus tested seed is the most effective way of controlling this virus.
- Chemical control of aphids is not an effective method for controlling non-persistently transmitted viruses, such as PSBMV.
- Pulse crops should be sown away from other legumes to minimise the spread of PSBMV.
Procedure followed at the centers in case of positive test
- Destroy seed, produce healthy seed
Referencs and further reading
Temperate Pulse Viruses: Pea Seedborne Mosaic Virus (PSBMV)
http://www.agls.uidaho.edu/ebi/vdie//vide/descr575.htm
 PSbMV (photos:www.dpi.vic.gov.au) |
Scientific name
Broad Bean Mottle Virus (BBMV)
Significance
The virus was seen as being of minor importance until reports of its widespread occurrence began to appear in the 1970s and 1980s.
Symptoms
Plants display leaf symptoms, dwarfing and production of few or no flowers. Infected plants are in concentric patches. BBMV can affect seed quality by causing necrosis and shrivelling of the seed. Seed transmission rates in legumes are low and range from 0.1% to 1.4%.
 Broad Bean Mottle (photo: www.padil.gov.au) |
Hosts
Under experimental conditions susceptibility to infection by virus is found in several families. Susceptible host species are found in the Family Amaranthaceae, Chenopodiaceae, Leguminosae-Papilionoideae, Solanaceae. The following species were susceptible to experimental virus infection: Chenopodium album, Chenopodium amaranticolor, Chenopodium hybridum, Chenopodium quinoa, Cyamopsis tetragonoloba, Glycine max, Gomphrena globosa, Lathyrus odoratus, Lens culinaris, Lupinus albus, Medicago sativa, Melilotus albus, Nicotiana clevelandii, Phaseolus vulgaris, Pisum sativum, Trifolium hybridum, Trifolium incarnatum, Trifolium pratense, Trifolium repens, Trifolium subterraneum, Vicia faba, Vicia villosa.
Geographic distribution
BBMV was reported in faba bean crops in Portugal, Sudan, Morocco, and Algeria. then undertook a regional survey and found BBMV in faba bean crops in Egypt, Morocco, Sudan, Syria and Tunisia. Fortass and Bos (1991) surveyed faba bean crops in Morocco and found that luteoviruses and BBMV were the most prevalent viruses.
Biology and transmission
BBMV survives between growing seasons of the primary pulse host in an alternative host or in infected seed. Due to the fact that the natural host range includes at least four families and includes many food legumes and non-legume wild hosts, it may survive in a range of alternative hosts.
Virus is transmitted by a vector. Virus is transmitted by mechanical inoculation; transmitted by grafting.
Virus is transmitted by arthropods, by insects of the order Coleoptera; Acalymma trivittata, Colaspis flavida, Diabrotica undecimpunctata, Sitona lineata.
Detection/indexing method in place at the CGIAR Center at ICARDA
- Tissue Blot Immuno Assay test (TBIA), Enzyme-linked immunosorbent assay (ELISA)
Treatment/control
- Seed is considered to be the main source of BBMV, therefore sowing virus tested seed is the most effective way of controlling this virus.
- Commercial seed tests are available.
- Chemical control of insects is not an effective method for controlling virus.
Procedure in case of positive test
- Destroying seed, producing healthy seed under controlled conditions
References and further reading
http://www.padil.gov.au/pbt/index.php?q=node/46&pbtID=152
http://www.ncbi.nlm.nih.gov/ICTVdb/ICTVdB/00.010.0.03.002.htm
Scientific name
Pea Early-Browning Virus(PEBV)
Other scientific name
Broad Bean Yellow Band Virus (BBYBV)
Significance
Not Significant.
Symptoms
The symptoms are systemic necrotic spots, streaks and mosaic, leaves malformed, plants stunted.
Hosts
Families containing susceptible hosts: Alliaceae, Chenopodiaceae, Compositae, or Cruciferae, Gramineae, Leguminosae-Papilionoideae, Solanaceae. Species inoculated with virus that do not show signs of susceptibility: Allium cepa, Avena sativa, Beta vulgaris, Brassica campestris ssp. rapa, Hordeum vulgare, Lactuca sativa, Lycopersicon esculentum, Medicago sativa, Nicotiana glutinosa, Solanum tuberosum, Trifolium incarnatum, Trifolium pratense, Trifolium repens, Triticum aestivum, Vicia faba.
Geographic Distribution
The virus occurs in Belgium, Italy, Morocco, the Netherlands, Sweden, and the United Kingdom.
Biology and transmission
Virus is transmitted by a vector. Virus is transmitted by mechanical inoculation; transmitted by seeds (37% in Pisum sativum cv. Rondo).
Vector Transmission: Virus is transmitted by nematodes; family Trichodoridae; Trichodorus teres, T. pachydermus in The Netherlands; T. anenome, T. primitivus, T. viruliferus
Detection/indexing method
- Not important (TBIA) Tissue Blot ImmunoAssay test, Enzyme-linked immunosorbent assay (ELISA)
Treatment/control
Not given.
Procedure followed at the centers in case of positive test
Not given
References and further reading
http://www.ncbi.nlm.nih.gov/ICTVdb/ICTVdB/00.072.0.01.002.htm
Scientific name
Red Clover Vein Mosaic Virus (RCVMV)
Other scientific name
Pea Stunt Virus
Significance
It reduces the yield of red clover by reducing the foliage growth, decreasing persistence and increasing susceptibility to root rots.
Symptoms
RCVMV is reported to cause stunting in faba beans and inoculated plants have been reported to show chlorotic lesions or general chlorosis, tip mottle and abscission.
Hosts
It also infects a number of temperate pulses including Cicer arietinum (chickpea), Lathyrus odoratus (sweet pea), Lens culinaris (lentil), Pisum sativum (field pea), and Vicia faba (faba bean, broad bean, tick bean). RCVMV is seedborne in Trifolium pratense (red clover), Pisum sativum and Vicia faba.
Geographic distribution
It occurs in North America, South Africa, Europe, and India.
Biology and transmission
- Virus is transmitted by a vector. Virus is transmitted by mechanical inoculation; transmitted by seeds (in Trifolium pratense and Vicia faba, not transmitted by pollen.
- Vector Transmission: Virus is transmitted by arthropods, by insects of the order Hemiptera, family Aphididae; Acyrthosiphon pisum, Myzus persicae, Therioaphis (Pterocallidium) maculatus, Cavariella aegopodii, C. theobaldi. Virus is transmitted in a non-persistent manner.
Detection/indexing method in place at the CGIAR Center at ICARDA
- Not important, (TBIA) Tissue Blot Immuno Assay test, Enzyme-linked immunosorbent assay (ELISA)
Treatment/control
- Use resistant varieties.
- Resistance to vectors is another approach to controlling viruses.
References and further reading
http://www.padil.gov.au/pbt/index.php?q=node/46&pbtID=172
http://www.ncbi.nlm.nih.gov/ICTVdb/ICTVdB/00.056.0.04.027.htm
http://www.apsnet.org/online/feature/plantdisease/text/fig08.htm
http://www1.agric.gov.ab.ca/$Department/deptdocs.nsf/All/prm7877?OpenDocument
Insects - faba bean
Contributors to this section: ICARDA, Syria (Siham Asaad, Abdulrahman Moukahal)
|
Contents: |
Scientific names
Bruchus rufimanus Boheman, 1833 & Bruchidius incarnatus Bohemann, 1833.
Significance
Beans may contain un-emerged pupae or adults. High levels of damage or infestation are unacceptable for export outlets for human consumption. For seed purposes, the damage may not be as serious, although germination of the seed may be slightly reduced.
Symptoms (Damage)
Damage to beans is characterized by a circular hole in the seed where the adult beetles emerge. Other types of damage such as seed staining and smaller holes may be seen where the larvae has not matured.
Hosts
Faba bean, cowpea, common bean.
Geographic distribution
Widespread in all varieties of winter and spring field beans and also broad beans.
Biology and transmission
Bruchus spp. adults emerge from hibernation in spring and after feeding on pollen and nectar lay eggs on the immature faba bean pods. The larvae bore a small round hole through the pod coat and enter the first avail able developing seed. Most of the larval development and pupation occurs in the hard seeds after harvest in the store. The mature larvae bite a hole from inside almost to the seed surface leaving only a transparent epidermal seed membrane, which are called ‘windows’. The adults remain in the seeds and only emerge through the ‘windows’ after sowing. If the seeds are not sown, but stored for another season the adults will die.
Detection/indexing method in place at the CGIAR Center at ICARDA
- Visual inspection
Treatment/control
- Since the infestation of Bruchus spp. starts in the field control methods should aim at reducing the population in the field before the larvae penetrate the seed.
-
Several cultural measures can be used to reduce Bruchus spp. infestations:
- Deep ploughing after harvest.
- Use of uninfected or treated seeds for sowing which has to be followed by all farmers to avoid immigration of the flying beetles from infested to uninfected fields.
- Storage of seeds for 2 successive seasons, so that all adults are dead at the time of sowing.
- Late sowing also reduced Bruchus spp. infestations. Because of the shorter growing period however, yields are also reduced. With regard to chemical control insecticides such as endosulfan can be applied in the field at early pod setting or harvested seeds can be fumigated with phosphine or methyl bromide. The annual routine fumigation conducted in Egypt decreased the infestation from more than 12% to less than 1%within 5 years.
- Screening of large numbers of germplasm for host plant resistance to B. incarnatus only revealed reduced infestation in the genotypes that are late in flowering and pod setting
Procedure followed at the centers in case of positive test
- Fumigation
References and further reading
http://www.inra.fr/hyppz/RAVAGEUR/6bruruf.htm
http://ressources.ciheam.org/om/pdf/a10/92605136.pdf
http://www.padil.gov.au/viewPestDiagnosticImages.aspx?id=394
 Bean Seed Beetle (photos: www.pgro.org) |
Cowpea seed beetle, Adzuki bean seed beetle
Scientific names
Callosobruchus maculatus (Fabricius), 1775, C. chinensis (L.), 1758.
Significance
The Callosobruchus beetles are principally a serious pest of stored products under hot dry conditions; complete destruction of grain and pulses may take place in a short time. In humid climates, the rates of increase of its competitors are so much greater that it has difficulty in establishing itself.
Symptoms (Damage)
The larval stage of the weevil tunnel and develop within the beans. They may consume nearly the entire bean contents. Pupation occurs in the beans and adults emerge through a round hole in the seed coat. Damage is a combination of the feeding and contamination.
Hosts
They attack several legumes in storage, chickpea (Cicer arietinum), arhar (Cajanus cajan), green gram (Vigna radiata), pea (Pisum sativum) and kidney bean (Phaseolus vulgaris) seeds. On lentil, C. chinensis is the most common.
Geographic distribution
Both species are widespread and found in all continents with subtropical or tropical conditions (USA, Mediterranean, India, and Australia).
Biology and transmission
The eggs are glued to the bean or the pod. On hatching the larvae bores into the seed where it makes a translucent 'window' in the seed before pupating. The larval and pupal stages are spent inside the bean. The adult emerges through the 'window' leaving a neat round hole. Infestations can begin in the field. Adults move to bean fields from trash beans left in sacks, harvesters, planters, or feed areas. The cowpea weevil readily attacks dried beans; thus this weevil can be a serious storage pest. Bean weevil infestations can also start in the field and may also originate from trash beans. As with the cowpea weevil, bean weevil will attack dried beans and can be a serious pest in stored beans. Broad bean weevil infestations also start in the field, but this pest is not a storage problem.
Detection/indexing method at ICARDA
- Visual inspection
Treatment/control
- Cleanliness in storage: Stores should be cleaned from all residues of grains, straw and flour and be de-infested by spraying walls with malathion, sacks, thresher and transportation vehicles also should be cleaned.
- Chemical control/protection: Seeds stored for food/feed can be fumigated with phospine (Phostoxin) available as 0.6 g pellets and 3 g tablets. The recommended dosage is 0.5-1 tablet/m3 with an exposure time of 2-4 and 3-5 days for pellets and tablets, respectively. The major advantages of Phostoxin are that it controls all insect stages, has no residues, does not affect flavor or germination and is easy to handle. Seeds stored for planting can be treated with insecticides such as Actellic at 4-10 ppm a.i. (0.5 g/kg seed) or malathion at 10 ppm a.i., which will protect the seeds for several months.
- Traditional methods of seed protection: Small quantities of seed stored for human consumption are often mixed with different vegetable oils or other substances. Mixing seeds with olive oil and salt (5 ml and 20 g/kg seed) or Neem seed oil (3 ml/kg seed) can provide adequate control for a period of 3-4 months.
Procedure followed in case of positive test
- Fumigation
References and further reading
http://www.icarda.org/Publications/Field_Guides/Lentil/Lentil.htm#Lent7.Html
http://www.infonet-biovision.org/default/ct/82/pests
http://agspsrv34.agric.wa.gov.au/Ento/pestweb/Query1_1.idc?ID=-1771861620
http://www.zin.ru/Animalia/coleoptera/rus/calmacdk.htm
http://www.uky.edu/~cfox/Students/Amarillo/Angela.html

Callosobruchus maculatus (photo: www.zin.ru/ ) |
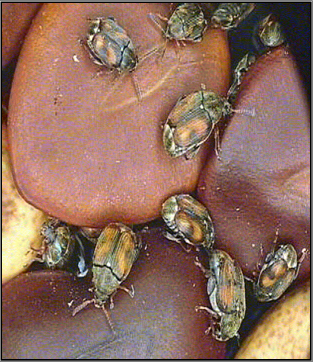
Callosobruchus maculatus (photo:www.uky.edu/) |

Callosobruchus chinensis |
Nematodes - faba bean
Contributors to this section: ICARDA, Syria (Siham Asaad, Abdulrahman Moukahal).
Stem and bulb nematode, Stem and bulb eelworm.
Scientific name
Ditylenchus dipsaci (Kühn, 1857) Filipjev, 1936.
Other scientific names
Tylenchus dipsaci (Kühn) Bastian, 1865. Ditylenchus phloxidis Kirjanova, 1951.
Ditylenchus fragariae Kirjanova, 1951.
Significance
D. dipsaci is one of the most devastating plant parasitic nematodes, especially in temperate regions. Without control, it can cause complete failure of host crops.
Symptoms
D. dipsaci causes swelling and deformation of stem tissue or lesions which turn reddish-brown then black, depending on cultivar and environmental factors. Newly formed pods take on a dark-brown appearance. The lesions envelop the stem and increase in length, often advancing to the edge of an internode. Leaf and petiole necrosis is also common under heavy infestations, but can be confused with symptoms induced by fungal leaf pathogens. Infected seeds are darker, distorted, and smaller in size and may have speckle-like spots on the surface. Heavy infestations often kill the main shoots, stimulating secondary tiller formation. The more severe symptoms are usually induced by the "giant race" on legumes.
Hosts
D. dipsaci is known to attack over 450 different plant species, including many weeds. However, it occurs in more than ten biological “races” some of which have a limited host-range. The race(s) that breed on rye, oats and onions seem to be polyphagous and can also infest several other crops, whereas those breeding on lucerne, Trifolium pratense and strawberries are virtually specific for their named hosts and seem to have relatively few alternative host plants. The tulip race will also infest Narcissus, whereas another race commonly found in Narcissus does not breed on tulip. It is known that some of the races can interbreed and that their progeny have different host preferences.
The principal hosts are faba beans, garlic, Hyacinthus orientalis, leeks, lucerne, maize, Narcissus pseudonarcissus, oats, onions, peas, Phlox drummondii, P. paniculata, potatoes, rye, strawberries, sugarbeet, tobacco, Trifolium pratense, T. repens, tulips. It has also been reported on carnations, celery, Hydrangea, lentils, rape, parsley, sunflowers, and wheat.
Geographic distribution
D. dipsaci occurs locally in most temperate areas of the world (Europe and the Mediterranean region, North and South America, northern and southern Africa, Asia and Oceania) but it does not seem able to establish itself in tropical regions except at higher altitudes that have a temperate climate. In most countries regulatory measures (e.g. certification schemes) are applied to minimize further spread of D. dipsaci.
Biology and transmission
In international trade D. dipsaci is liable to be carried on dry seeds and planting material of host plants. In the field the fourth-stage juvenile can withstand desiccation for many years, and although soil densities seem to decrease rapidly, the nematode can survive for years without a host plant. Nematode survival and damage are greater in heavy soils as compared to sandy soils. It can also survive on a number of weeds. Irrigation water and cultivation by contaminated farm tools and machinery are other sources of inoculum dissemination.
Detection/indexing method at ICARDA
- Nematode extraction test
Treatment/control
- Control by crop rotation is limited by the polyphagous habit of some races of D. dipsaci and by persistence of the nematode in clay soils. Chemical treatments of the soil are not an economic proposition for large areas. However, it may sometimes be worth treating small patches, after lifting and destroying the affected plants (bulbs) together with a margin of surrounding healthy ones, to eradicate a slight infestation before it spreads.
- Nematode-free (certified) seeds and planting material are most essential to prevent crop damage by D. dipsaci. Hot-water treatments with different temperature-time combinations, depending on type and state of seed material, are operational and efficient to control D. dipsaci. Systemic nematicides may be effective to some extent Ditylenchus dipsaci in controlling D.dipsaci in some ornamental crops. The use of tolerant or resistant cultivars can also reduce the damage.
- In laboratory experiments faba bean bulbs were heat treated in an attempt to control Ditylenchus dipsaci. A wide range of temperatures (43 to 50 deg C) and treatment times (30, 60, 120 min) was tested but none was successful. Chemical treatment, particularly preplanting treatment, was more successful. Calcium polysulphide (8%) and nemaphos (0.1 and 0.2%) were very effective.
Procedure followed at the centers in case of positive test
- Fumigation, rotation.
References and further reading
http://www.eppo.org/QUARANTINE/nematodes/Ditylenchus_dipsaci/DITYDI_ds.pdf
http://www.doacs.state.fl.us/pi/enpp/nema/nemacirc/nem187.pdf
http://www.ipmimages.org/browse/detail.cfm?imgnum=0162066
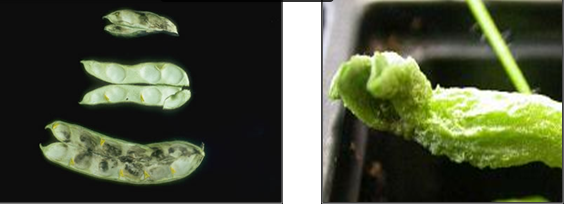 Stem and bulb nematode (photos:www.ipmimages.org/) |
More Articles...
- Weeds - faba bean
- Best practices for the safe transfer of faba bean germplasm
- Risk management
- Risk assessment
- Risk identification and analysis
- Risk evaluation
- Decision support tool
- Guide for users
- Evaluating the Cost-Effectiveness of Collection Management: A Methodological Framework
- Overview: Testing tissue-culture material of clonal crops for pests
Subcategories
-
main
- Article Count:
- 11
-
Stog
- Article Count:
- 2
-
Stog-rice
- Article Count:
- 7
-
Stog-sorghum
- Article Count:
- 11
-
Stog-common-bean
- Article Count:
- 10
-
stog-forage-legume
- Article Count:
- 10
-
stog-forage-grass
- Article Count:
- 11
-
stog-maize
- Article Count:
- 9
-
stog-chickpea
- Article Count:
- 10
-
stog-millets
- Article Count:
- 12
-
stog-barley
- Article Count:
- 10
-
stog-groundnut
- Article Count:
- 9
-
stog-pigeon-pea
- Article Count:
- 8
-
stog-wheat
- Article Count:
- 10
-
stog-lentil
- Article Count:
- 9
-
stog-cowpea
- Article Count:
- 10
-
stog-faba-bean
- Article Count:
- 9
-
risk management
- Article Count:
- 4
-
decision support tool
- Article Count:
- 3
-
stog-clonal
- Article Count:
- 23
-
developing strategies
- Article Count:
- 4




